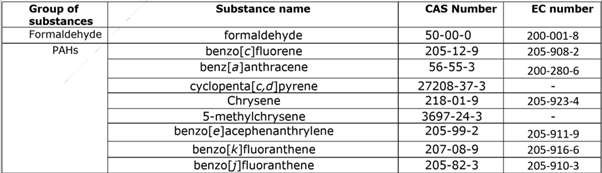
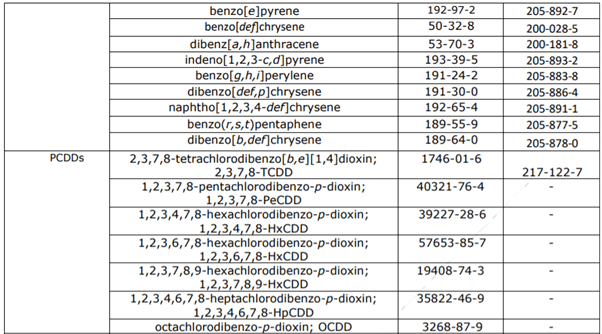
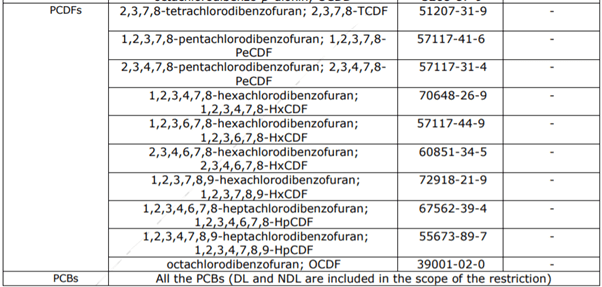
The non-exhaustive list also consists of formaldehyde, dioxins, furans, PCBs as well PAHs, and other substances. This restriction was grounded on Composition 69 (1) of the REACH Regulation. It aims to reduce the health threat associated with the operation of single-use baby diapers.
The substances included in this restriction are listed below :
A urine simulator was used to descry and quantify substances using realistic logical tests.
The procedures performed a result that the quantum of PAH, PCDD/ F, PCB, and/ or formaldehyde used in baby diapers isn’t controlled duly.
So that the Risk Management Options (RMO) carried out several analyses to find a way to borrow the proposed restriction in a further doable way and concluded in REACH restriction.
The restrictions are as below:
| Substances | Conditions of the restriction |
|---|---|
| Formaldehyde (CAS Number: 50-00-0)
Polychlorobiphenyls (DL-PCBs and NDLPCBs) The PAHs, PCDDs, PCDFs, and PCBs involved in this restriction are listed in table 1. |
1. Shall not be placed on the market, after 01/01/2024, in any of the disposable baby diapers such as: • Traditional baby diapers, o Diaper pants, or training pants for toilet training the child, 2. For the entire articles listed in paragraph 1, the following substances should not migrate in a concentration equal to or greater than the migration limits specified below: i. Formaldehyde in individual migration limit equal to or greater than 0.42 mg/kg of diaper for all the entire articles specified in paragraph 1 3. Paragraphs 1 to 2 shall apply without prejudice to the application of any stricter restrictions or existing regulations. 4. Paragraphs 1 to 2 shall not apply to i. Re-usable diapers. 5. An analytical method developed using extraction by urine simulant in a whole diaper shall be used as the test method for demonstrating the conformity of articles to paragraphs 1 and 2. A standardized method needs to be defined. The restriction shall apply 24 months after it enters into force.
|
Reach out to our regulation experts on product regulatory compliances

